Nominee 2006
Fokussiertes Laserlicht
An instrument ideally suited to this purposed was developed by Karin Schütze, Carsten Hoyer and Yilmaz Niyaz of P.A.L.M. Microlaser Technologies GmbH in Bernried. Karin Schülze is head of innovation management at the company; Carsten Hoyer manages the department Development & Science. Yilmaz Niyaz is an application specialist.
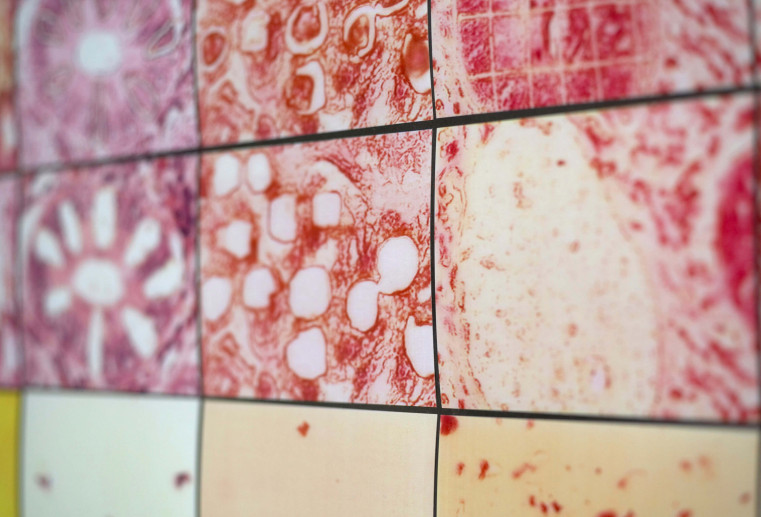
High purity is decisive
When doing research on genes and proteins, it essential that all specimens are as pure as possible. Only pure specimens provide indisputable analyses and allow individual molecules to be classified to certain cell functions – in order to be able to identify the mechanisms involved in the development of diseases.
more details
Resumes

Dr. rer. nat. Karin Schütze
- 24.04.1956
- geboren in Heilbronn
- 1975 – 1985
- Studium: Biologie und Sport für das Lehramt an der Ruprecht-Karls-Universität, Heidelberg; Promotion am Institut für Angewandte Physikalische Chemie, Promotionsprüfung an der Fakultät für Pharmazie, Ruprecht-Karls-Universität, Heidelberg
- 1985 – 1989
- Laborleitung am Institut für Angewandte Physikalische Chemie in Heidelberg: Aufbau und Organisation des Zellbiologielabors im BMFT-Projekt LABIO (Laseranwendungen in der Biologie). Experimente und Weiterentwicklung der UV-Laser-Mikrostrahlapparatur
- 1989 – 1990
- Feodor-Lynen-Fellow der Alexander-von-Humboldt-Stiftung an der Universität von Kalifornien in Berkeley. Aufbau und Experimente mit der Infrarot-Laserfalle am Beckman-Laser-Institut (Universität von Kalifornien in Irvine) und am AT&T Bell Laboratorium (Holmdel, New Jersey): Aufbau einer Infrarot-Laserpinzette in Berkeley in Zusammenarbeit mit Dr. Art Ashkin; AT&T Bell Laboratorium.
- 1991 – 2001
- Leiterin des Applikativen Laserzentrums im Krankenhaus Harlaching, München
- 1993
- Mitbegründerin der P.A.L.M. Microlaser Technologies GmbH, Wolfratshausen
- 1993 – 2000
- Berater der Firma P.A.L.M. Microlaser Technologies GmbH, Wolfratshausen
Verantwortungsbereiche Forschung, Entwicklung, wissenschaftliches Marketing, Vertrieb - 2000 – 2001
- Leitung der Forschung & Entwicklung (F&E) bei P.A.L.M. Microlaser Technologies GmbH, Bernried
- 2001 – 2005
- Direktorin F&E und erweiteter Vorstand bei P.A.L.M. Microlaser Technologies AG in Bernried
Entwicklung der Geräteplattform sowie Erforschung innovativer Applikationen; Suche nach neuen Märkten, Initiierung und Leitung - 2002 – 2005
- Mitglied des Aufsichtsrats bei P.A.L.M. Microlaser Technologies AG, Bernried
- 2004
- Verkauf des Unternehmens an die Carl Zeiss AG
- seit 2005
- Leitung des Innovationsmanagements bei P.A.L.M. Microlaser Technologies GmbH, Bernried
Weitere Tätigkeiten:
- seit 2003
- Mitglied im Programmausschuss „Optische Technologien“ des Bundesministeriums für Bildung und Forschung (BMBF)
- seit 2003
- Rotarierin im RC Weilheim
- seit 2005
- Interessenvertretung der europäischen Technologieplattform „Photonics21“
- seit 2006
- Mitglied im Kuratorium der Bayern-Innovativ GmbH
Ehrungen:
- 1984
- Auszeichnung der Staatsexamensarbeit mit einem Sonderpreis der Walther und Christine Richtzenhain Stiftung des Deutschen Krebsforschungszentrums, Heidelberg
- 1998
- Innovationspreis der Bayerischen Volksbanken und Raiffeisenbanken für die P.A.L.M. GmbH als „Mittelstandsbetrieb des Jahres 1998“
- 2001
- Philip-Morris-Forschungspreis
- 2002
- Anerkennungspreis im Rahmen des Bayerischen Innovationspreises
- 2006
- Berthold Leibinger Innovationspreis

Dr. rer. nat. Carsten Hoyer
- 15.09.1965
- geboren in Mannheim
- 1985 – 1992
- Studium der Chemie an der TU Karlsruhe und an der Ruprecht-Karls-Universität, Heidelberg
- 1992 – 1993
- Diplomarbeit am Institut für Physikalische Chemie an der Ruprecht-Karls-Universität, Heidelberg
- 1993 – 1996
- Dissertation am Institut für molekulare Biotechnologie an der Friedrich-Schiller-Universität, Jena
- 1997
- Promotionsprüfung an der Biologisch-Pharmazeutischen Fakultät der Friedrich-Schiller-Universität, Jena
- 01.07.1997
- Produktmanager für Inverse Mikroskope Carl Zeiss Jena GmbH
- 2001 – 2004
- Leitung des Bereichs BioMed im Produktmanagement des Geschäftsbereichs Lichtmikroskopie beim Carl Zeiss Werk, Göttingen
- 2004 – 2005
- Mitglied des Aufsichtsrats bei P.A.L.M. Microlaser Technologies GmbH, Bernried
- seit 2005
- Leiter der Abteilung Development & Science bei P.A.L.M. Microlaser Technologies GmbH, Bernried
Mitgliedschaften:
- seit 2002
- Vertreter von Carl Zeiss Göttingen im Measurement Valley e. V.

Dr. rer. nat. Yilmaz Niyaz
- 18.07.1969
- geboren in Istanbul/Türkei
- 1991 – 1992
- Studium der Biologie im Vordiplom an der Freien Universität Berlin, Berlin
- 1992 – 1998
- Studium der Biologie an der Ruprecht-Karls-Universität Heidelberg und Diplom-Abschluss
- 1997 – 2002
- Wissenschaftlicher Mitarbeiter, Ruprecht-Karls-Universität, Heidelberg
- 2003
- Promotion in Biologie an der Ruprecht-Karls-Universität, Heidelberg
- 2003 – 2005
- Applikationsspezialist im Bereich Entwicklung bei der Fa. P.A.L.M. Microlaser Technologies AG, Bernried, im Bereich Forschung & Entwicklung
- seit 2005
- Applikationsspezialist im Applikationslabor bei der Fa. P.A.L.M. Microlaser Technologies GmbH, Bernried
Contact
Spokesperson
Dr. rer. nat. Karin Schütze
Leitung Innovationsmanagement
P.A.L.M. Microlaser Technologies GmbH
Ein Unternehmen der Carl Zeiss MicroImaging Gruppe
Am Neuland 9 + 12
82347 Bernried
Tel.: +49 (0) 8158 / 99 71 214
Fax: +49 (0) 8151 / 99 71 149
E-Mail: karin.schuetze@palm-microlaser.com
Web: www.palm-microlaser.com
Press
Sieglinde Hinteregger
P.A.L.M. Microlaser Technologies GmbH
Ein Unternehmen der Carl Zeiss MicroImaging Gruppe
Am Neuland 9 + 12
82347 Bernried
Tel.: +49 (0) 8158 / 99 71 218
Fax: +49 (0) 8158 / 99 71 249
E-Mail: s.hinteregger@palm-microlaser.com
Web: www.palm-microlaser.com
Gudrun Vogel
Carl Zeiss Jena GmbH
Kommunikation
Carl-Zeiss-Promenade 10
07745 Jena
Tel.: +49 (0) 3641 / 64 27 70
Fax: +49 (0) 3641 / 64 29 41
E-Mail: g.vogel@zeiss.de
Web: www.zeiss.de
A description provided by the institutes and companies regarding their nominated projects
From the concept to international technology prominence
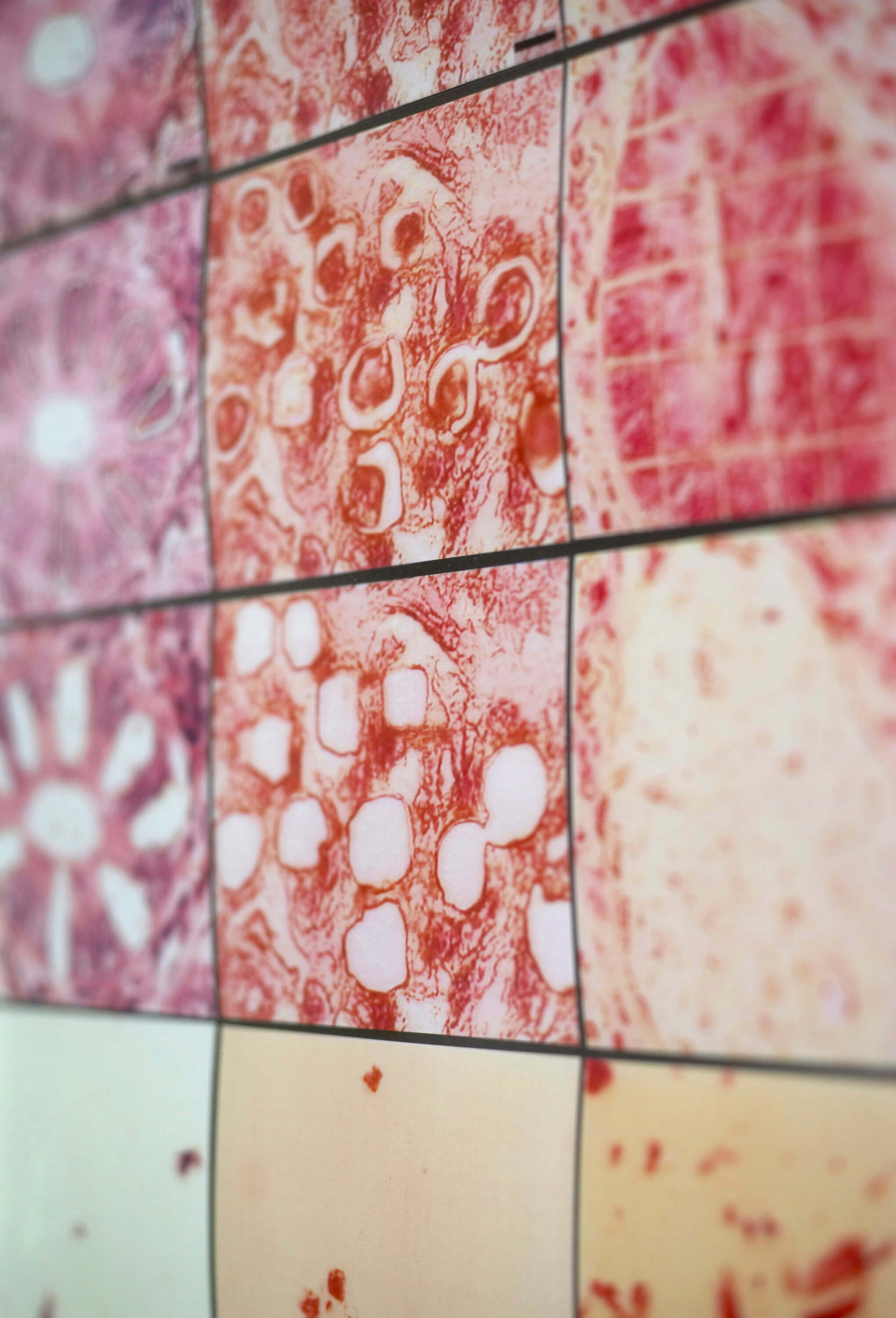
Biotechnology and biophotonics have become increasingly important over the past few years both for fundamental research in the area of molecular biology and medicine as well as for in the applying of obtained findings to the treatment of disease, products or active substances. One of the most important tasks in this area is the accurate explanation of cellular processes at the molecular level. These processes lead to genetic cytomorphosis and thus to diseases such as cancer. Until recently, medical diagnostics relied on the eye and experience of the pathologist. The newly developed and established molecular-biological methods, in contrast, give detailed evidence that can be used directly in diagnosis and treatment.
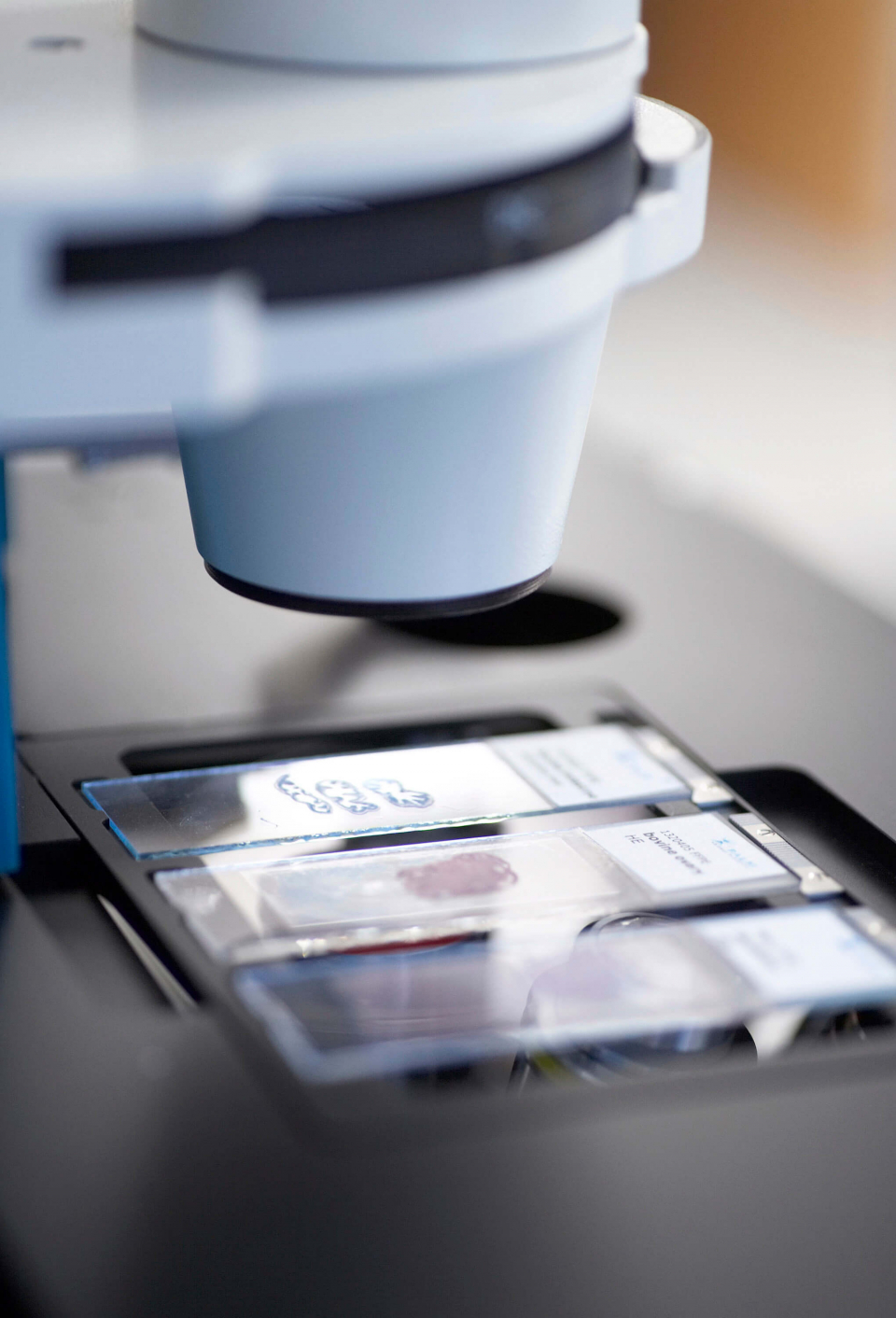
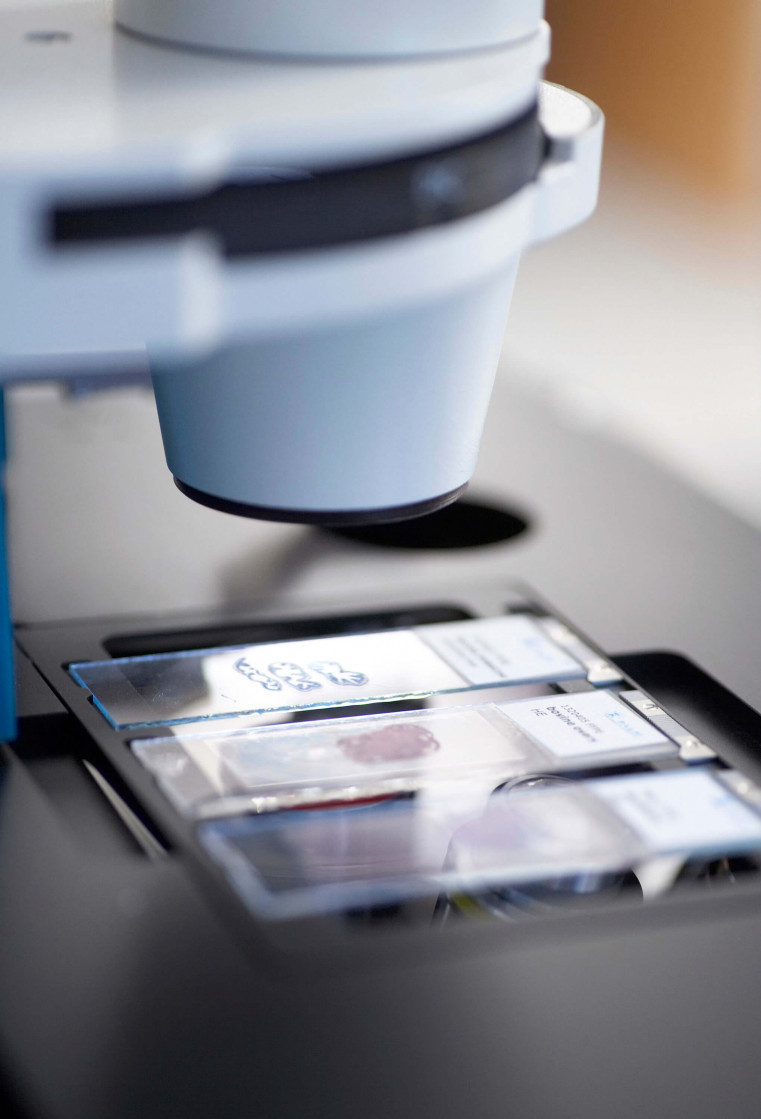
Yet the molecular causes of cell processes can only then be revealed if the samples studied are pure. This means that they may not consist of a mixture of healthy and diseased cells. A consistent and convincing analysis and diagnosis can only be prepared with uncontaminated, pure sample material since that is the only way results can be reliably classified and a specific therapy initiated.
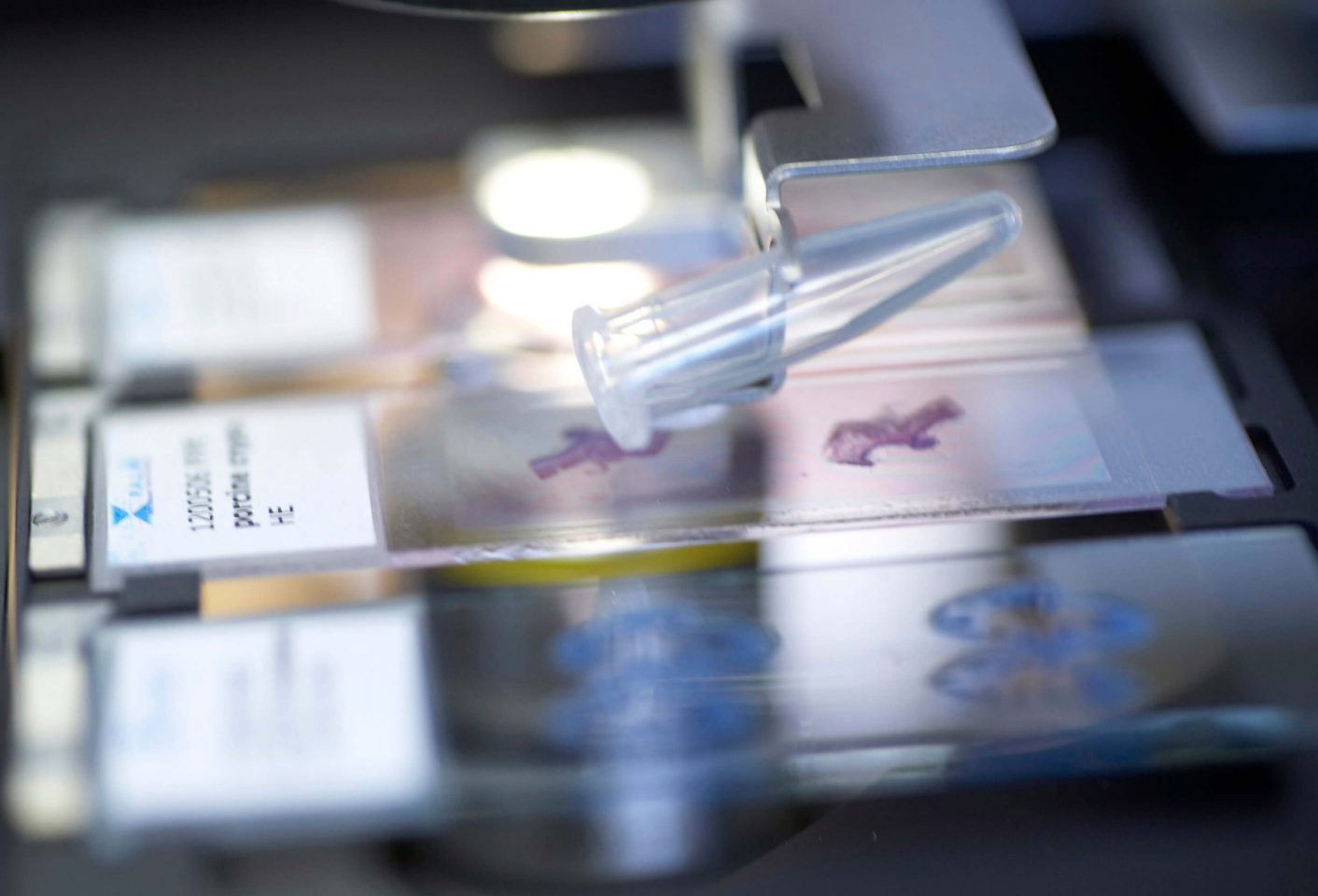
Consequently, non-contact preparation procedures of individual cells play an important role in the future of biomedicine. One of these procedures, in the meantime in use in labs around the world, was developed by the company P.A.L.M. in 1993 and is being marketed internationally by this technology leader. Without any contact, and thus entirely eliminating the possibility of contamination, a laser beam cuts individual cells or small cell areas from a tissue sample, ejects it out of the preparation level based solely on the power of the bundled light, and transports it directly into a sample vessel. This procedure combines “cutting” and “catapulting” and is known by its abbreviation LMPC, Laser Microdissection and Pressure Catapulting.
The PALM Technology
Bundled laser beams focused by means of a microscope lens to a diameter of ca. 1 µm make it possible to capture and move as well as the highly precise micro-manipulation of cells or sub cellular structures as well as of non-biological materials. Depending on the laser type used, there are two different principles of laser micro manipulation.
For the OpticalTweezers, a continuous laser that works in the red and near-infrared range is coupled with a research microscope and focused through a lens with a high numerical aperture. The resulting beam forces allow microscopically small objects to be captured, trapped, and moved. In this way, moving organisms, for example, such as bacteria or sperm can be trapped or single individuals be selected from a mixed population for subsequent molecular-biological analysis.
In the laser micro manipulation system PALM MicroBeam, a pulsed ultraviolet-A laser is coupled with the microscope and focused to a few micrometers through the lens. Such high energy densities are produced in the laser’s point of focus that biological material can be broken down or cut into individual molecules. This phenomenon is based on so-called ablative photo-decomposition, a photochemical process, in which all chemical bonds are broken by the extreme photon density in the narrowly focused laser. This happens within fractions of nanoseconds so that no transfer of heat to directly adjacent regions can occur. All molecules outside of the area of the laser’s focus remain unchanged. Contamination is avoided.
Originally, mechanical aids, such as needles or capillaries were used to extract sampled specimens. During laser dissecting work, it was discovered by accident that lasers can be used not only for freeing samples without cutting, but also to catapult samples out of the cells. To do so, the laser is focused slightly beneath the sample, the cut parts are ejected from the object level by means of the resulting wave of pressure directly into the capture vessel. The combination of “laser cutting and laser catapulting” was patented under the term LMPC. In the meantime, it has developed into an entirely non-contact, reliable, and fast laser preparation method. Several hundred publications on the subject of laser micro dissection and laser catapulting have shown that the process in no way damages DNA and RNA nor proteins. The PALM MicroBeam equipment system has become an integral part of molecular biological research and analysis.
Areas of Application
The LMPC method is fast becoming a key technology of modern biosciences and molecular medicine: it gives functional genetic and protein research entirely new insight into cellular mechanisms and understanding of the development of disease. Pure samples are a must in biomedical research to produce consistent molecular-biological analyses, unequivocally identify functional connections, or rapidly and accurately diagnose dysfunctions. The areas of application for laser micro dissection are thus varied and range from isolating individual chromosomes or cell compartments to the harvesting of selected cells from fixed tissue samples to patient samples such as sputum, cytosmears, or biopsy materials. It is also possible to harvest living cells from cell cultures for subsequent cloning. Even entire organisms survive laser-based transport.
The PALM MicroBeam system is not only widely used in tumor research, but is increasingly also making inroads in botany for investigating the processes and interactions in plant cells. The very latest and most interesting applications are in forensics where, for example, laser-isolated saliva, hair, and sperm samples or skin cells are catapulted directly from the evidence tape, thus without contact, into the sample vessel of the crime lab and are thus available for determining the “genetic fingerprint” for conviction of the perpetrator. Another interesting prospect is the isolating and “cleaning up” of living cells in stem cell research. Even extremely sensitive stem cells can be catapulted without losing their unique properties so that differentiated and non-differentiated stem cells can be purposely isolated and cultivated. Selected cell clones constitute the basis for stem cell research and therapy or for tissue cultivation in regenerative medicine. Uncontaminated living cell populations are increasingly indispensable also in the development and testing of specific ingredients in personalized medicine. This has resulted in incredible savings in terms of time in drug testing.
The full potential of laser micro manipulation has yet to be exhausted. Above all, research on cellular responses to active substances could play an important role in patient-specific treatments. The combination of laser micro-manipulation such as microinjection or cell fusion with the catapult technology to selectively sort successfully manipulated cells is a challenge and with a corresponding degree of automation offers innovative applications for stem cell research and regenerative medicine.
The PALM Systems
The current generation of PALM systems consists of an inverse research microscope (ZEISS Axiovert), a laser of a specific wavelength with a selected beam quality and a software package developed specifically for laser micro manipulation for automatic sampling. A special microscope stage carries several slides or sample vessels in microtiter format with excellent precision, and an innovative RoboMover sorts catapulted samples in up to 96 individual vessels on a microtiter plate. Image analysis software helps users locate the relevant cells.
Company History
The company P.A.L.M. was established by Karin and Raimund Schütze in 1993 as a GmbH (= a public limited company) with domicile in a private home in Wolfratshausen. The success of the company is primarily due to the synergies of technical knowledge, a feeling or patience for accuracy and the courage to pursue alternative solutions combined with user-oriented research and development. A master goldsmith, Raimund Schütze was responsible for development and production as well as installation and service of the laser microscope systems, while Karin Schütze was accountable for user-oriented research, the system design, and marketing (scientific marketing and sales). The first systems were developed and built in the basement. The hobby room served as the research and test lab, the children’s room functioned as an office, the kitchen as a meeting room, and final assembly took place in the living room.
Constant growth as a consequence of intense research work, publications in trade journals, and a presence at scientific symposiums led to an increase in personnel and an expansion in terms of space so that in 1998 the company moved to a new building in Bernried on Lake Starnberg south of Munich together with 10 employees. Two years later the company had 24 employees. A separate lab was set up at P.A.L.M. for molecular analysis of micro-dissected samples. P.A.L.M. GmbH sought investors to support corporate growth and became a stock company in August 2000. In the meantime, several hundred PALM systems are installed worldwide in research labs and university clinics. P.A.L.M. Microlaser Technologies GmbH now employs 60 employees. Sales of PALM systems are handled by the Carl Zeiss sales network and other international dealers. A team of highly qualified and committed employees provides excellent customer service and sales support, reliable production and service as well as innovative system and application development.
P.A.L.M. was and is part of several state-funded research projects with the goal of providing users innovative solutions for trend-setting tasks – an important prerequisite to keep and develop a position as the international technology leader. P.A.L.M. AG has belonged to the Carl Zeiss group since September 2004. P.A.L.M. Microlaser Technologies GmbH is now a one hundred percent subsidiary of Carl Zeiss MicroImaging GmbH.
The right to nominate outstanding achievements for the Deutscher Zukunftspreis is incumbent on leading German institutions in science and industry as well as foundations.
The project Non-contact harvesting of biological specimens for research and diagnostics was nominated by the Philip Morris Research prize.


 Gebärdensprache
Gebärdensprache
 Leichte Sprache
Leichte Sprache